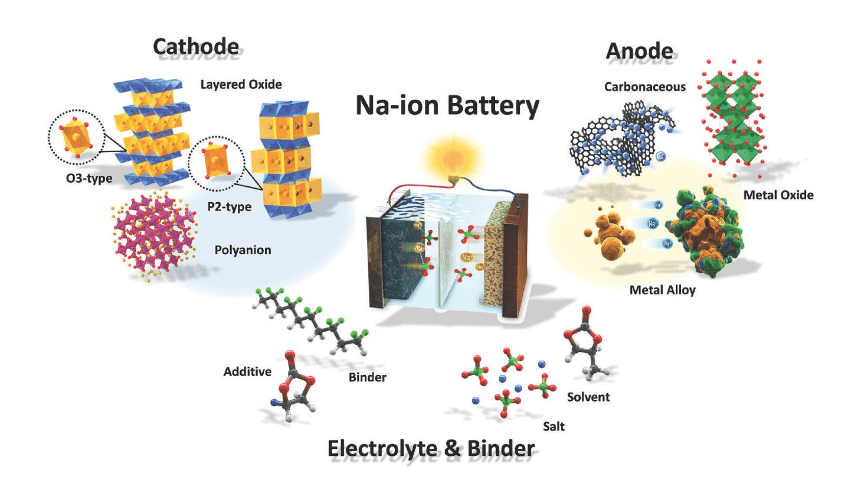
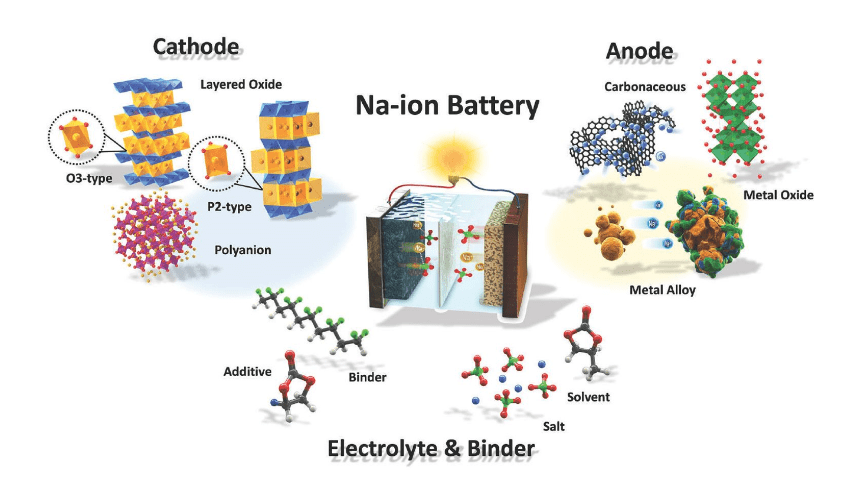
Calorimetry plays a crucial role in enhancing the safety and performance of sodium-ion batteries (SIBs) by providing detailed thermal characterization essential for understanding their operational stability. This method allows researchers to assess heat generation during battery operation, which is critical for preventing thermal runaway and improving overall efficiency.
What are calorimetric methods and why are they important for sodium-ion batteries?
Calorimetric methods involve measuring the heat produced or absorbed during chemical reactions, such as those occurring in sodium-ion batteries. These methods are vital because they provide insights into thermal behavior, which is essential for predicting battery performance and safety. By understanding how heat is generated during charging and discharging cycles, researchers can identify potential risks associated with overheating and develop strategies to mitigate them.
Chart: Overview of Calorimetric Techniques Used in Battery Research
| Technique | Description | Application |
|---|---|---|
| Differential Scanning Calorimetry (DSC) | Measures heat flow associated with phase transitions | Evaluating thermal stability |
| Tian-Calvet Calorimetry | Provides precise heat flow measurements | Analyzing heat generation during cycling |
How does thermal characterization contribute to battery safety?
Thermal characterization helps identify critical temperature thresholds at which materials may begin to decompose or react exothermically. Understanding these thresholds allows engineers to design safer batteries by selecting materials that can withstand higher temperatures without degrading. Additionally, it aids in developing effective cooling strategies that can prevent overheating during operation.
What insights can calorimetric measurements provide about sodium-ion batteries?
Calorimetric measurements offer valuable insights into the electrochemical processes occurring within sodium-ion batteries. For example, they can reveal how different electrode materials interact with electrolytes at various temperatures, which is crucial for optimizing battery chemistry. Furthermore, these measurements help predict the state-of-health (SOH) of batteries by monitoring changes in heat generation patterns over time.
Chart: Heat Generation Profiles During Cycling
| Cycle Stage | Heat Generation (J) | Observations |
|---|---|---|
| Charging | 1.3 | Lower heat generation observed |
| Discharging | 1.5 | Higher heat generation noted |
| Cycle Aged | Increased | Indicates potential degradation |
What are the implications of thermal stability for sodium-ion battery technology?
The thermal stability of sodium-ion batteries is critical for their commercial viability. High thermal stability reduces the risk of thermal runaway, a condition where excessive heat leads to catastrophic failure. By improving the materials used in SIBs—such as selecting more thermally stable electrolytes—researchers can enhance safety while maintaining performance.
How do sodium-ion batteries compare to lithium-ion batteries in terms of safety and performance?
Sodium-ion batteries offer several advantages over lithium-ion counterparts:
- Lower Flammability: Sodium-based systems generally exhibit lower flammability compared to lithium systems, reducing fire risks.
- Thermal Stability: SIBs can operate safely at higher temperatures due to their inherent material properties.
- Cost Efficiency: Sodium is more abundant and less expensive than lithium, potentially lowering production costs.
However, SIBs currently have lower energy density compared to lithium-ion batteries, which may limit their application in high-performance scenarios like electric vehicles.
Chart: Comparison of Sodium-Ion and Lithium-Ion Batteries
| Feature | Sodium-Ion Batteries | Lithium-Ion Batteries |
|---|---|---|
| Energy Density | ~150 Wh/kg | ~250 Wh/kg |
| Flammability | Lower | Higher |
| Cost | Lower due to abundant materials | Higher due to scarcity |
| Thermal Stability | Higher | Moderate |
What future trends are anticipated in sodium-ion battery research and development?
Future trends in sodium-ion battery research include:
- Material Innovations: Continued exploration of new cathode and anode materials that enhance energy density and cycle life.
- Advanced Electrolytes: Development of non-flammable electrolytes that improve safety without sacrificing performance.
- Integration with Renewable Energy: Increased focus on using SIBs for grid storage applications, enabling better integration of renewable energy sources.
As research progresses, these advancements could lead to broader adoption of sodium-ion technology across various applications.
Latest News
Recent studies highlight significant advancements in sodium-ion battery technology through innovative calorimetric techniques. Researchers have successfully demonstrated how these methods enhance understanding of thermal behavior, leading to safer and more efficient designs. As companies invest in SIB technology, experts predict a growing market presence alongside traditional lithium-ion systems, particularly for applications requiring lower costs and enhanced safety.
Editor Comment
“The integration of calorimetry into the development process of sodium-ion batteries marks a pivotal shift towards safer energy storage solutions,” says Dr. Ijaz Ul Mohsin, a leading researcher in battery technology. “By understanding the thermal dynamics at play, we can create more reliable systems that meet the demands of modern energy applications.”
FAQ Section
Q: What is calorimetry?
A: Calorimetry is a technique used to measure the amount of heat generated or absorbed during chemical reactions, crucial for understanding battery performance.Q: Why are sodium-ion batteries considered safer than lithium-ion batteries?
A: Sodium-ion batteries typically exhibit lower flammability and higher thermal stability compared to lithium-ion systems, reducing risks associated with overheating.Q: How does thermal stability affect battery life?
A: Improved thermal stability minimizes the risk of thermal runaway, enhancing overall safety and prolonging battery life by preventing material degradation.Q: What advancements are being made in sodium-ion technology?
A: Research focuses on developing new materials and electrolytes that improve energy density while maintaining safety standards.